Poster:
Stability Meets Scalability
Breakthrough Upstream AAV Manufacturing with a Novel Transfection Complex Stabilizer Additive
Summary
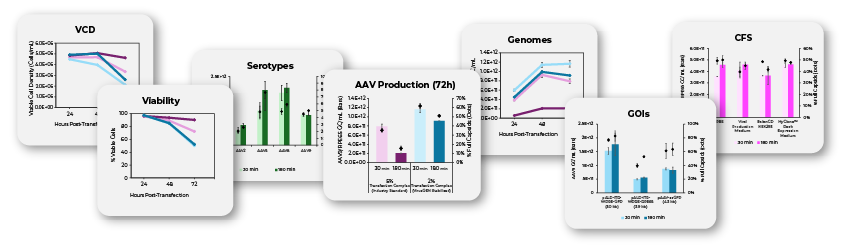
In the rapidly advancing field of cell and gene therapy, manufacturing efficiency continues to be a hurdle for advancing AAV therapeutics from clinical development to commercial-scale production. Two pressing issues impacting AAV manufacturing scale-up are the limited stability of transfection complexes and the considerable volumes these complexes require—issues that become significantly challenging at commercial scale, restrict upstream production workflows, and create time pressures within GMP manufacturing environments. To overcome these limitations, Mirus Bio has developed the VirusGEN® Transfection Complex Stabilizer, an innovative transfection additive specifically engineered to simultaneously address both challenges and streamline processes while scaling to large-scale bioreactors. This novel solution dramatically extends transfection complex viability from minutes to hours while enabling manufacturers to reduce complex volume requirements from the conventional 5% to only 2% of total culture volume. This reduction in volume requirement delivers particular value in large-scale operations seeking to expand into 1,000 L bioreactors or beyond, while yielding substantial benefits in material consumption, preparation time, and operational complexity. We here share data obtained from a collaborator highlighting how VirusGEN® Transfection Complex Stabilizer resulted in extending the stability of prepared transfection complex to up to 2 hours and reducing the volume requirement to 2%. By providing both extended stability and reduced volume, VirusGEN® Stabilizer makes large-scale AAV bioreactor operations more feasible, eliminating typical workflow constraints while maintaining robust AAV yields during scale-up.
Access Research On:
- AAV Manufacturing Challenges
- How to Stabilize Transfection Complex Size
- How to Achieve Robust Stabilization at 5% and 2% Transfection Complexes
- Compatibility with Multiple Serotypes, GOIs, and Complex Formation Buffers
- Consistency with Pumping
- Scalability in terms of VCD, Viability, Genomes, and Production
- Reagent Comparison Conducted by Forge Biologics