TransIT®-Lenti Transfection Reagent – 5 x 1.5 mL
Ideal transfection reagent for recombinant lentivirus production
$2,754.00

Unsure of what transfection reagent you need? Consult Reagent AgentTM.
Related Products
-

TransduceIT® Transduction Reagent – 1 mL
$34.00 Add to cart -

TransIT®-Lenti Transfection Reagent – 0.3 mL
$160.00 Add to cart -

TransIT®-Lenti Transfection Reagent – 0.75 mL
$399.00 Add to cart -

Lentivirus Packaging Mix – 5 rxn
$522.00 Add to cart -

TransIT® Lentivirus System – 5 rxn
$588.00 Add to cart -

TransIT®-Lenti Transfection Reagent – 1.5 mL
$634.00 Add to cart -

Lentivirus Packaging Mix – 34 rxn
$2,089.00 Add to cart -

TransIT® Lentivirus System – 34 rxn
$2,207.00 Add to cart -

TransIT®-Lenti Transfection Reagent – 10 x 1.5 mL
$5,066.00 Add to cart
Description
TransIT®-Lenti Transfection Reagent is designed to enhance delivery of packaging and transfer vectors to adherent HEK 293T and suspension 293-F cell types to increase recombinant lentivirus production.
The TransIT® Lentivirus System combines the benefits of TransIT®-Lenti with the Lentivirus Packaging Mix Powered by MISSION® Genomics to produce even higher functional titers.
- High Performance – Provide higher functional titers
- Simple Protocol – No media change required, single harvest
- Animal Origin Free – Regulatory friendly
SKU: MIR 6605
Supporting Data
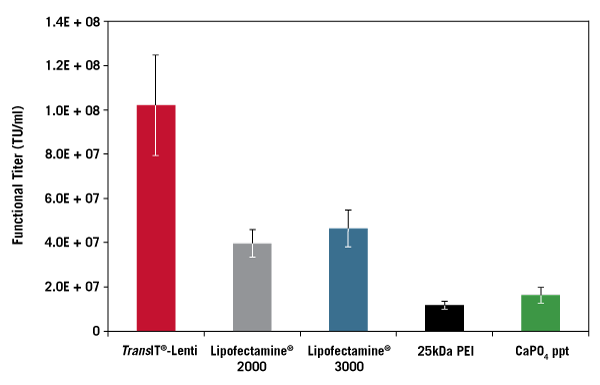
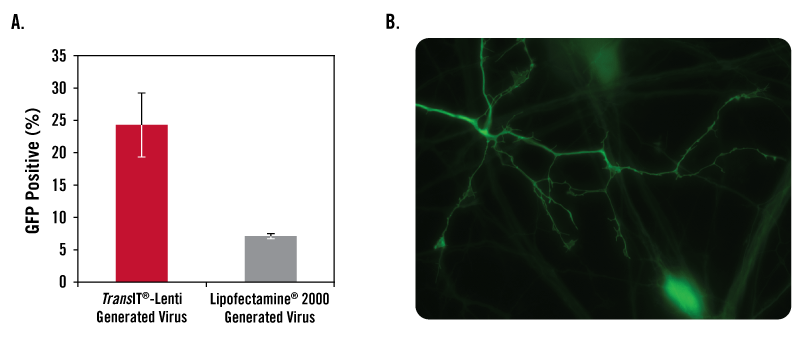
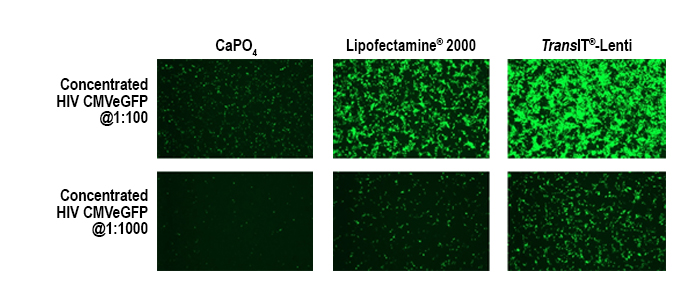
Data courtesy of Jeremy Coffin, University of Iowa Viral Vector Core
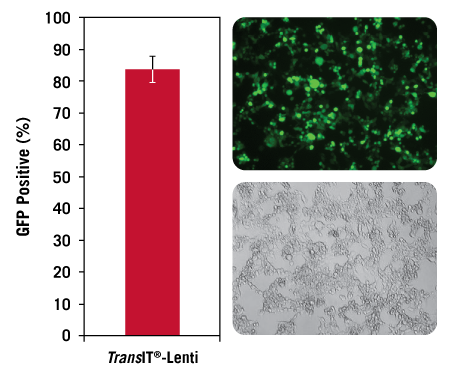
High Efficiency Transfection with TransIT®-Lenti Transfection Reagent. Adherent 293T/17 cells were transfected in a 6-well plate format using the pLKO.1-puro-CMV-TurboGFP™ transfer vector and the Lentivirus Packaging Mix Powered by MISSION® Genomics using the TransIT®-Lenti Transfection Reagent (3:1, vol:wt). GFP efficiency was measured at 48 hours post-transfection using guava easyCyte™ 5HT Flow Cytometer. Error bars represent five transfection complexes. Images were captured at 48 hours post-transfection (10X objective) using a Zeiss Axiovert S100 inverted fluorescence microscope. The observed cell rounding and cell-cell fusion is due to high expression of the vesicular stomatitis virus G protein (VSV-G) for pseudotyping the recombinant lentivirus.
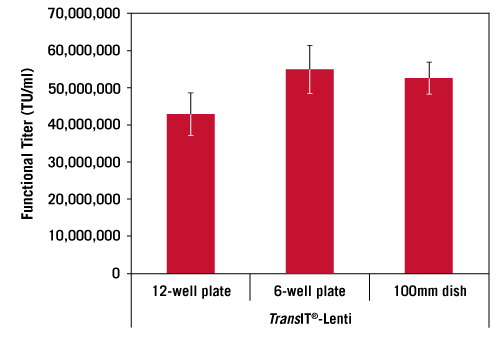
Lentivirus Production is Scalable. Adherent 293T/17 cells were transfected in a 12-well, 6-well or 100 mm plate format using the pLKO.1-puro-CMV-TurboGFP™ transfer vector and the Lentivirus Packaging Mix Powered by MISSION® Genomics at a 1:1 ratio, and the TransIT®-Lenti Transfection Reagent (3:1, vol:wt). The supernatant was harvested, filtered (0.45 µm), and titered using 293T/17 cells. Lentivirus transductions were performed in the presence of 8 µg/ml TransduceIT™ and GFP expression was measured 72 hours post-transduction using guava easyCyte™ 5HT Flow Cytometer. Error bars represent triplicate transfection complexes titered individually. Functional titers were calculated using virus dilutions with less than 20% GFP positive cells.
Resources
Specifications
Storage Conditions
TransIT®-Lenti Transfection Reagent: Store at -20°C
Lentivirus Packaging Mix Powered by MISSION® Genomics: Multiple storage conditions – see individual bottles for specific recommendations
TransIT® Lentivirus System: Store at -20°C
TransduceIT™ Transduction Reagent: Store at -20°C
Product Guarantee
TransIT®-Lenti Transfection Reagent: 1 year
Lentivirus Packaging Mix Powered by MISSION® Genomics: 1 year
TransIT® Lentivirus System:
TransduceIT™ Transduction Reagent: 6 months
Usage Statement
All Configurations: For Research Use Only.
Animal Origin Statement
All Configurations: This product is animal origin free.
Technical Product Literature
Full Protocols
TransIT® Lentivirus System Full Protocol (PDF)
TransIT®-Lenti Full Transfection Protocol (PDF)
Quick Reference Protocols
TransIT® Lentivirus System Quick Ref Protocol (PDF)
TransIT®-Lenti Quick Ref Protocol (PDF)
Product Data Sheets
Lentivirus Packaging Mix Product Data Sheet (PDF)
TransduceIT™ Reagent Product Data Sheet (PDF)
SDS
Lentivirus Packaging Mix SDS (PDF)
TransduceIT™ Reagent SDS (PDF)
TransIT® Lentivirus System SDS (PDF)
TransIT®-Lenti SDS (PDF)
FAQs
See the TransIT-Lenti FAQs
Using transfection reagents and enhancers from Mirus has given our platform a competitive advantage. Increasing efficiency and lowering costs for all of our programs.
Sally Mader, PhD
ZYZ Theraputics


