Applications | High Throughput Transfection
- High Throughput
- Reverse Transfection of DNA
- RNAi Screening Protocol
- HT Citations and Reagents
- Products
High Throughput Transfection
Advances in high throughput (HT) screening as well as availability of multi-well cDNA/shRNA/siRNA/miRNA libraries have made high throughput transfection of different nucleic acids commonplace. High throughput transfections can be carried out in multi-well formats (such as 96-well, 384-well, etc.) using reverse transfection protocols that are amenable to automated robotic systems.

Forward Transfection
The most routinely employed transfection protocol where cells are seeded a day prior to transfection is referred to as “forward transfection”. Forward transfection methods work well for most adherent cell types that are seeded a day prior to transfection in order to achieve an actively dividing cell population at the time of transfection. A typical forward transfection protocol using TransIT-X2® Dynamic Delivery System can be found here (PDF).
Reverse Transfection
For high throughput applications, a “reverse transfection” protocol, where freshly passaged cells are added to pre-plated transfection complexes is ideal as it reduces overall experimental time for the end user. Cell culture time can be further reduced by using frozen assay-ready cells for some experiments. Reverse transfections are also compatible with most automated robotic systems.

Typical Reverse Transfection Protocol Workflow. On the day of transfection, complexes are prepared in a multiwell plate and incubated for the recommended complex formation time. Freshly passaged cells are then directly added to the multiwell plates containing the transfection complexes at twice the density of a standard forward transfection protocol. This reduces hands-on time for the end-user by one day compared to forward transfection and makes the protocol amenable to liquid handling.
Modified Reverse Transfection
In “modified reverse” transfections, cells are passaged and plated immediately before transfection complexes are added to the cells. In this scenario, adherent cells are loosely adhered to the plate surface by the time they interact with the transfection complexes.
Solid Phase Reverse Transfection
If the nucleic acid to be transfected is immobilized or spotted in a multi-well format already as in the case of cDNA/shRNA/siRNA screens, the transfection protocol is referred to as “solid phase reverse transfection”.
Reverse Transfection Protocol for DNA
All TransIT® DNA transfection reagents, including TransIT-X2® Dynamic Delivery System, can be used for reverse transfection of DNA into multiple cell types. Reverse transfection is the transfer of genetic material into cells and is “reverse” because the order of DNA and cells is reverse that of conventional transfection (Wikipedia). Additionally, cryopreserved cell stocks can be utilized for immediate transfection reducing overall culture time.
Reverse Transfection Protocol for DNA in 96-well Plates Using TransIT-X2® Dynamic Delivery System
An example of high efficiency forward and reverse transfection of A549 cells using the broad spectrum TransIT-X2® Dynamic Delivery System can be found in the figure below.
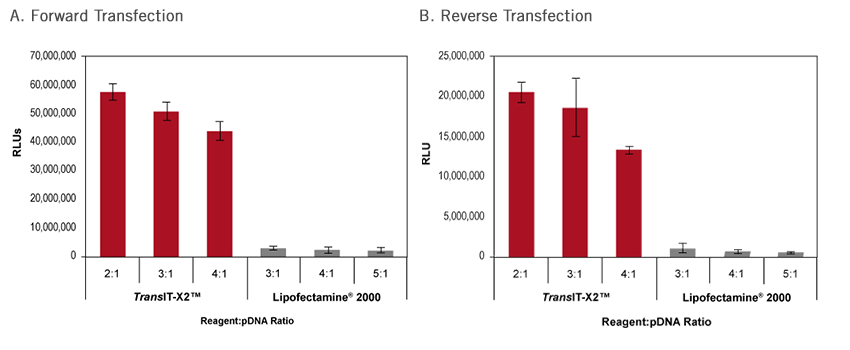
Comparison of Forward and Reverse Transfection in A549 Cells Using TransIT-X2® Dynamic Delivery System. A549 cells were seeded 24 hours prior to transfection for forward transfection (A). For reverse transfection (B), A549 cells were plated after adding complexes following the protocol below. Cells were transfected with a firefly luciferase encoding plasmid DNA (0.1 µg DNA) using either TransIT-X2® Dynamic Delivery System or Lipofectamine® 2000 Transfection Reagent at the indicated reagent-to-DNA ratios in a 96-well plate for both forward and reverse transfection protocols. Luciferase activity was measured at 24 hours post-transfection using a conventional luciferase assay. Both forward and reverse transfection using TransIT-X2® Dynamic Delivery System yield superior luciferase expression compared to Lipofectamine® 2000.
Before You Start
Akin to forward transfection, optimizing transfection conditions by transfecting a reporter plasmid into the cell type of interest (such as a luciferase or GFP encoding plasmid) into cells is critical prior to high throughout screening.
- For best results, determine the appropriate dose of DNA and transfection reagent in the multi-well format for the screen. For example, the optimal DNA concentration generally ranges between 0.05-0.2 μg/well of a 96-well plate with an optimal TransIT-X2®:DNA ratio of 2-6 μl per 1 μg DNA.
- Observe reporter gene expression and toxicity at regular time points over at least a 24 hour period, optimally for 48 hours. Use the optimal DNA and transfection reagent dosage for HT screening applications.
A. Cell Plating Prior to Transfection
- At least 24 hours prior to transfection, plate cells at an appropriate cell density in a T-75 cm2 flask or similar tissue culture dish so that the cells will be 70-80% confluent the following day. Approximately 2-6 x 106 cells will be needed per 96-well plate. Multiple flasks may need to be prepared if more than one 96-well plate is to be transfected.
- Incubate the cells overnight.
B. Complex Formation for 96-well Plates
- In each well of the 96-well plate to be used for transfection, add appropriate amount of serum-free medium (i.e. Opti-MEM® I Reduced Serum Medium) (see Table 1). Note: Alternatively, a mastermix can be prepared in a sterile tube if transfecting the same plasmid throughout the plate. Calculations are shown in Table 1 for 120 wells to account for pipetting errors. If a transfection master mix is prepared, prepare the transfection mixture as follows (Steps 1-4) and add per well to the 96-well plate using a multi-channel pipettor or liquid handler after complex formation (Step 4).
- Add appropriate amount of stock plasmid DNA (see Table 1) to the wells containing the each well of the 96-well plate.
- Add appropriate amount of TransIT-X2® to the Opti-MEM® I plasmid DNA mixture (see Table 1).
- Incubate at room temperature for at least 15 minutes to allow for the transfection complexes to form. Note: If you are using pre-printed cDNA/shRNA screens, allow for an additional 10 minutes for the reconstitution of the dried printed DNA. Do not allow complexes to incubate longer than 60 minutes before adding cells from step C below.
C. Cell Plating in 96-well Plates
- Trypsinize cells in T-75 cm2 flask (from Step A) as per standard tissue culture procedure. Note: To prevent re-adherence of the cells to the flask, perform this step no more than one hour prior to transfection. To further reduce cell culture time, cell plating can also be performed using cryopreserved cell stocks, after they are centrifuged to remove DMSO and counted using Trypan Blue.
- Add 5-10 ml of complete media to the cell suspension. Mix thoroughly by pipetting.
- Count the cells using a hemacytometer to determine the appropriate volume of cells in media to obtain 1.6-4.8 x 105 cells per ml.
- Add 92 µl of diluted cell mixture (1.4-4.4 x 104 cells) to each well. Gently rock the dish back and forth and from side to side to distribute the cells evenly. Do not swirl or rotate the dish, as this may result in uneven distribution.
- Incubate 24-48 hours.
- Harvest and assay for gene expression or other reporter assay.
High Throughput RNAi Screening Protocol
Increased availability of synthetic siRNA screens in the recent years has positioned multiwell RNAi screening as a powerful technique for understanding gene function. Delivery of smaller nucleic acids such as siRNA and miRNA is relatively straightforward as delivery to the cytoplasm is sufficient to initiate gene knockdown. Mirus offers three different reagents for siRNA or miRNA transfection – TransIT-X2® Dynamic Delivery System, TransIT-TKO® and TransIT-siQUEST®. Any of these three reagents can be used for reverse transfecting short species of RNA such as siRNA and miRNA into a broad range of cell types. TransIT-X2® Dynamic Delivery System exhibits high knockdown with either forward or reverse transfection; see data below followed by a detailed reverse transfection protocol using TransIT-X2® Dynamic Delivery System in a 96-well format.
Reverse Transfection Protocol for siRNA in 96-well Plates Using TransIT-X2® Dynamic Delivery System
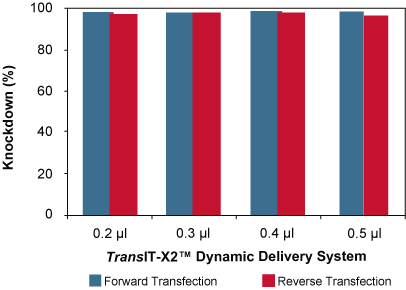
High Knockdown Using TransIT-X2® System for Forward or Reverse Transfection in CHO Cells Stably Expressing Firefly Luciferase. For the forward transfection (blue), CHO-luc cells were seeded in 96-well plates 24 hours prior to transfection; whereas, for reverse transfection (red), cells were plated after adding complexes following the protocol below. Cells were transfected with 25 nM of either a non-targeting siRNA or a anti-firefly luciferase siRNA in complex with different amounts of TransIT-X2® Dynamic Delivery System (indicated beneath each bar). Luciferase expression, normalized to non-targeting siRNA control were measured at 24 hours post-transfection. Using either forward or reverse transfection, high knockdown is observed with a broad range of TransIT-X2®:siRNA ratios.
Before You Start
Optimizing knockdown conditions in the cell type of interest by transfecting a RNAi reporter (such as a luciferase targeting siRNA) before conducting an HT RNAi screen is critical, if allowed by the experimental set-up. Alternatively, siRNAs targeting a non-essential endogenous gene can be employed. There are also options to directly track siRNA delivery by using labeled RNAi controls such as Label IT® RNAi Delivery Controls. A comprehensive resource on additional considerations for high throughout RNAi screening such as siRNA specificity and controlling off-target effects can be found here (1).
- Determine the appropriate dose of siRNA (10-50 nM final concentration per well) and TransIT-X2® (0.2-0.6 µl) per well of the 96-well format to be used for HT RNAi screening.
- Observe reporter knockdown and toxicity at regular time points over at least a 48 hour period, optimally for 72 hours.
A. Cell Plating Prior to Transfection
- At least 24 hours prior to transfection, plate cells at an appropriate cell density in a T-75 cm2 flask or similar tissue culture dish so that the cells will be 70-80% confluent the following day. Approximately, 2-6 x 106 cells will be needed per 96-well plate. Multiple flasks may need to be prepared if more than one 96-well plate is to be transfected.
- Incubate the cells overnight.
B. Complex Formation for 96-well Plates
- In each well of the 96-well plate to be used for transfection, add appropriate amount of serum-free medium (i.e. Opti-MEM® I Reduced Serum Medium) (see Table 1). Note: Alternatively, a mastermix can be prepared in a sterile tube if transfecting the same siRNA throughout the plate. Calculations are shown in Table 1 for 120 wells to account for pipetting errors. If a transfection master mix is prepared, prepare the tansfection mixture as follows (Steps 1-4) and add per well to the 96-well plate using a multi-channel pipettor or liquid handler after complex formation (Step 4).
- Add appropriate amount of stock siRNA (see Table 1) to the wells containing the each well of the 96-well plate.
- Add appropriate amount of TransIT-X2® to the Opti-MEM® I siRNA mixture (see Table 1).
- Incubate at room temperature for at least 15 minutes to allow for the transfection complexes to form. Note: If you are using preprinted siRNA screens, allow for an additional 10 minutes for the reconstitution of the dried printed siRNA. Do not allow complexes to incubate longer than 60 minutes before adding cells as per Step C below.
C. Cell Plating in 96-well Plates
- Trypsinize cells in T-75 cm2 flask (from Step A) as per standard tissue culture procedure. Note: To prevent re-adherence of the cells to the flask, perform this step no more than one hour prior to transfection. To further reduce cell culture time, cell plating can also be performed using cryopreserved cell stocks, after they are centrifuged to remove DMSO and counted using Trypan Blue.
- Add 5-10 ml of complete media to the cell suspension. Mix thoroughly by pipetting.
- Count the cells using a hemacytometer to determine the appropriate volume of cells in media to obtain 1.6-4.8 x 105 cells per ml.
- Add 92 µl of diluted cell mixture (1.4-4.4 x 104 cells) to each well. Gently rock the dish back and forth and from side to side to distribute the cells evenly. Do not swirl or rotate the dish, as this may result in uneven distribution.
- Incubate 24-48 hours.
- Harvest and assay for knockdown of gene expression or other reporter assay.
High Throughput Citations and Transfection Reagents
TransIT® Transfection Reagents have been used successfully by various researchers in high throughput applications in multi-well formats ranging from 96-well to 384-well (as mentioned in the Citations Table below).
Citations for Reverse Transfection using TransIT® Transfection Reagents
| Citation | Cell Type Transfected | Nucleic Acid Transfected | TransIT® Reagent Used | Robotic system used | Multi-well format | Application |
|---|---|---|---|---|---|---|
| Wollenick et al. Nucleic Acids Res. 2012, 40 (5) 1928-1943 | U2OS | Plasmid DNA | TransIT®-LT1 | None | 96-well | Solid phase reverse transfection using a 96-well genome-wide full length cDNA array |
| Owens et al. J Biol Chem. 2010 February 26; 285(9): 6761-6769. | HeLa | Plasmid DNA | TransIT®-LT1 | Multidrop 384 (Titan) | 384-well | HT cell-based screens to detect stabilized protein targets following chemical mutagenesis |
| Zhao et al. Molecular Neurodegeneration 2009, 4:4 | HeLa | Plasmid DNA | TransIT®-LT1 | None | 384-well | HT fluorescence polarization-based Aβ degradation assay |
| Warzecha et al. Mol Cell. 2009 March 13; 33(5): 591-601. | HEK 293T cell clone stably expressing the luciferasesplicing reporter | Plasmid DNA | TransIT®-293 | Wellmate Handler (Matrix) | 384-well | HT cell-based genome-wide cDNA expression screening |
| Moser et al. PLoS ONE 2013 8(6):e68129 | HeLa | siRNA | TransIT-TKO® | Hydra DT robot | 96-well | HT genome-wide siRNA screen to identify host factors necessary for growth of the parasite Toxoplasma gondii |
| Lu et al. Oncogene. 2011 November 10; 30(45): 4567-4577. | MDA-MB-468 | siRNA | TransIT-TKO® | None | 96-well | HT kinome siRNA phosphoproteomic screen using an siRNA library targeting 541 kinases and kinase-related genes |
| Choudhary et al. J Biol Chem 2011 October, 286, 37187-37195. | A549-Luc stable reporter cell line | siRNA | TransIT-siQUEST® | Titertek, multidrop 384 cell dispenser | 96-well | HT siRNA screening of the human kinome |
| Andersen et al. Mol Ther. 2010 November; 18(11): 2018-2027. | Human mesenchymal stem cells (hMSCs) | siRNA | TransIT-TKO® | None | Tissue culture plates coated by a lyophilization process with TransIT-TKO®/siRNA particles | Tissue engineering using siRNA coated nanostructured scaffolds |
| Liu et al. J Biol Chem. 2008 August 22; 283(34) 23169-23178. | Ikkγ-/- MEFs | siRNA | TransIT-siQUEST® | None | Not specified | Mechanism study for RSV infection |
All of the TransIT® transfection reagents can be used for reverse transfections with the following general recommendations based on the nucleic acid to be delivered. All of these are broad spectrum formulations are chemically distinct. Depending on the cell type being transfected, one reagent may have superior performance over others; for cell-type specific recommendations, please consult the Reagent Agent® transfection database.
Broad Spectrum HT Transfection Reagent Recommendations
| Nucleic Acid to be Transfected | HT Transfection Reagent Recommendation |
|---|---|
| Plasmid DNA and siRNA | TransIT-X2® Dynamic Delivery System |
| Plasmid DNA including cDNA and shRNA encoding plasmid DNA libraries | Option 1: TransIT-X2® Dynamic Delivery System Option 2: TransIT®-2020 Transfection Reagent Option 3: TransIT®-LT1 Transfection Reagent |
| siRNA/miRNA | Option 1: TransIT-X2® Dynamic Delivery System Option 2: TransIT-TKO® Transfection Reagent Option 3: TransIT-siQUEST® Transfection Reagent |
| Large mRNA | TransIT®-mRNA Transfection Kit |
| Oligonucleotides | TransIT®-Oligo Transfection Reagent |
Products for High Throughput Transfection
All of the TransIT® transfection reagents can be used for reverse transfections with the following general recommendations based on the nucleic acid to be delivered. All of these are broad spectrum formulations are chemically distinct. Depending on the cell type being transfected, one reagent may have superior performance over others; for cell-type specific recommendations, please consult the Reagent Agent® transfection database.
Broad Spectrum High Throughput Transfection Reagent Recommendations
| Nucleic Acid to be Transfected | High Throughput Transfection Reagent Recommendation |
|---|---|
| Plasmid DNA and siRNA | TransIT-X2® Dynamic Delivery System |
| Plasmid DNA (including cDNA and shRNA encoding plasmid DNA libraries) | Option 1: TransIT-X2® Dynamic Delivery System Option 2: TransIT®-2020 Transfection Reagent Option 3: TransIT®-LT1 Transfection Reagent |
| siRNA/miRNA | Option 1: TransIT-X2® Dynamic Delivery System Option 2: TransIT-TKO® Transfection Reagent Option 3: TransIT-siQUEST® Transfection Reagent |
| Large mRNA | TransIT®-mRNA Transfection Kit |
| Oligonucleotides | TransIT®-Oligo Transfection Reagent |
Technical Resources
![]() Don’t See Your Cell Type? Consult Reagent Agent® Transfection Database
Don’t See Your Cell Type? Consult Reagent Agent® Transfection Database
Citation Database: Check if our reagents have been used by other researchers to transfect your cell type
Technical Support: Speak directly with a transfection expert