AAViator™
Production
Platform
Deliver the Productivity the Industry Requires
- Out-of-the-box performance
- Multiple serotypes, cell lines, and scales
- Support to speed evaluation process
Achieve viral titer
greater than
1e12 gc/mL
across serotypes


AAViator™
Production
Platform
Deliver the Productivity the Industry Requires
- Out-of-the-box performance
- Multiple serotypes, cell lines, and scales
- Support to speed evaluation process
Achieve viral titer
greater than
1e12 gc/mL
across serotypes


AAViator™
Production
Platform
Deliver the Productivity the Industry Requires
- Out-of-the-box performance
- Multiple serotypes, cell lines, and scales
- Support to speed evaluation process
Achieve viral titer
greater than
1e12 gc/mL
across serotypes


| Experimental Condition | AAViator™ Production Platform | Polymer F | Polymer P | Lipid A Kit |
|---|---|---|---|---|
| Cells | Commercially Available HEK293 cells 2.0 | |||
| Growth Medium | Cellvento® 4HEK | Commercially Available AAV Production Medium | ||
| Complexation Medium | DMEM High Glucose | Lipid A Complexation Buffer | ||
| Transfection Reagent | TransIT® AAViator | Polymer F | Polymer P | Lipid A Transfection Reagent |
| Enhancer | RevIT™ AAV Enhancer | N/A | Lipid A Enhancer | |
| Supplement | N/A | Lipid A Booster | ||
Our Challenge:
Getting Your Therapeutic to Patients
Maximized Performance
Out-of-the-box productivity with less optimization to achieve key milestones from bench to bioreactor.


Commercial Readiness
Begin with the end in mind.
Straightforward quality & documentation.
Economic process viability.
Speed to Clinic
Reduced time spent on optimization, scale-up and accelerated process development.


Our Challenge:
Getting Your Therapeutic to Patients
Maximized Performance
Out-of-the-box productivity with less optimization to achieve key milestones from bench to bioreactor.


Commercial Readiness
Begin with the end in mind.
Straightforward quality & documentation.
Economic process viability.
Speed to Clinic
Reduced time spent on optimization, scale-up and accelerated process development.


Our Challenge:
Getting Your Therapeutic to Patients
Maximized Performance
Out-of-the-box productivity with less optimization to achieve key milestones from bench to bioreactor.


Commercial Readiness
Begin with the end in mind.
Straightforward quality & documentation.
Economic process viability.


Speed to Clinic
Reduced time spent on optimization, scale-up and accelerated process development.
Multifold Gains in Upstream Productivity

Thaw and Expand Cells
Medium and Inoculum Prep
Virus Production
Cell Growth and Transfection

Cell Lysis and DNA Digestion
Viral Vector Purification Formulation and FF
Formulation and FF
Upstream Improvements in Titer and % Full Capsids
Downstream
Fill/Finish
Enabling Next-Generation AAV Production
Get the Data
Gains in upstream yield and quality reduce pressure on downstream purification operations.
See the methodology and results that confirm the benefits of the AAViator™ Production Platform as a ready-to-use solution for upstream AAV processing.
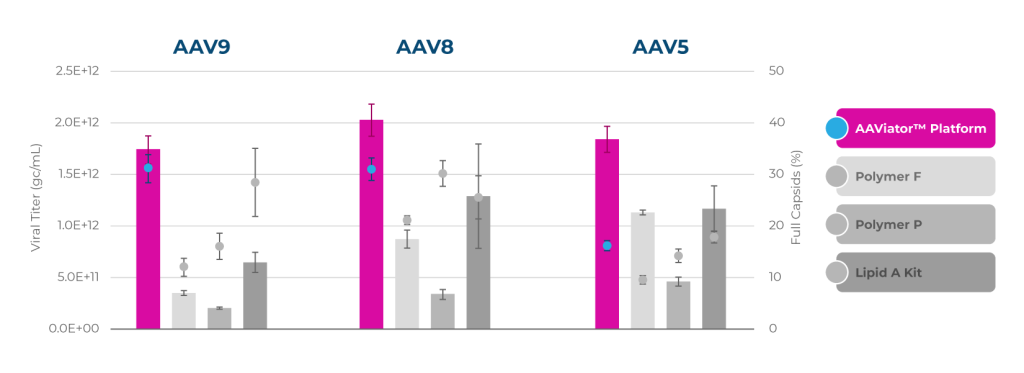
2-9X higher titers and percentages of full capsids across serotypes
2-5X higher titers and percentages of full capsids across cell lines
Packaging efficiency across serotypes
Consistent 1.2×1012 GC/mL & 30-50% full capsids at scale

Get the Data
Gains in upstream yield and quality reduce pressure on downstream purification operations.
See the methodology and results that confirm the benefits of the AAViator™ Production Platform as a ready-to-use solution for upstream AAV processing.
Your Gene Therapy’s Cohesive Support
Single point of support for transfection and upstream process optimization.
Avoid challenges requiring multiple touchpoints and coordination with disparate stakeholders
Performance Summary
Out-of-the-box performance requiring less optimization to achieve key milestones.
Proven performance and versatility across multiple AAV serotypes, HEK 293 cell lines, and production scales.
Straightforward protocol speeds evaluation process. Access protocols and technical documents.

Performance Summary
Out-of-the-box performance requiring less optimization to achieve key milestones.
Proven performance and versatility across multiple AAV serotypes, HEK 293 cell lines, and production scales.
Straightforward protocol speeds evaluation process. Access protocols and technical documents.

Speed to Clinic
Reduce time spent on optimization, scale-up, and accelerated process development with an Evaluation Kit or work with a Field Application Scientist to set up your gene therapy for long term success.
Read the White Paper →
Or